News
Latest News

E.W. Scripps Folding Scripps News, Eliminating 200 Jobs; Stock Jumps 15%
By Jon Lafayette published
Scripps News president Kate O’Brian leaving company

Hoda Kotb To Step Down From ‘Today’ Co-Anchor Post
By Michael Malone published
She joined the NBC morning show in 2007, and will stay on into early 2025
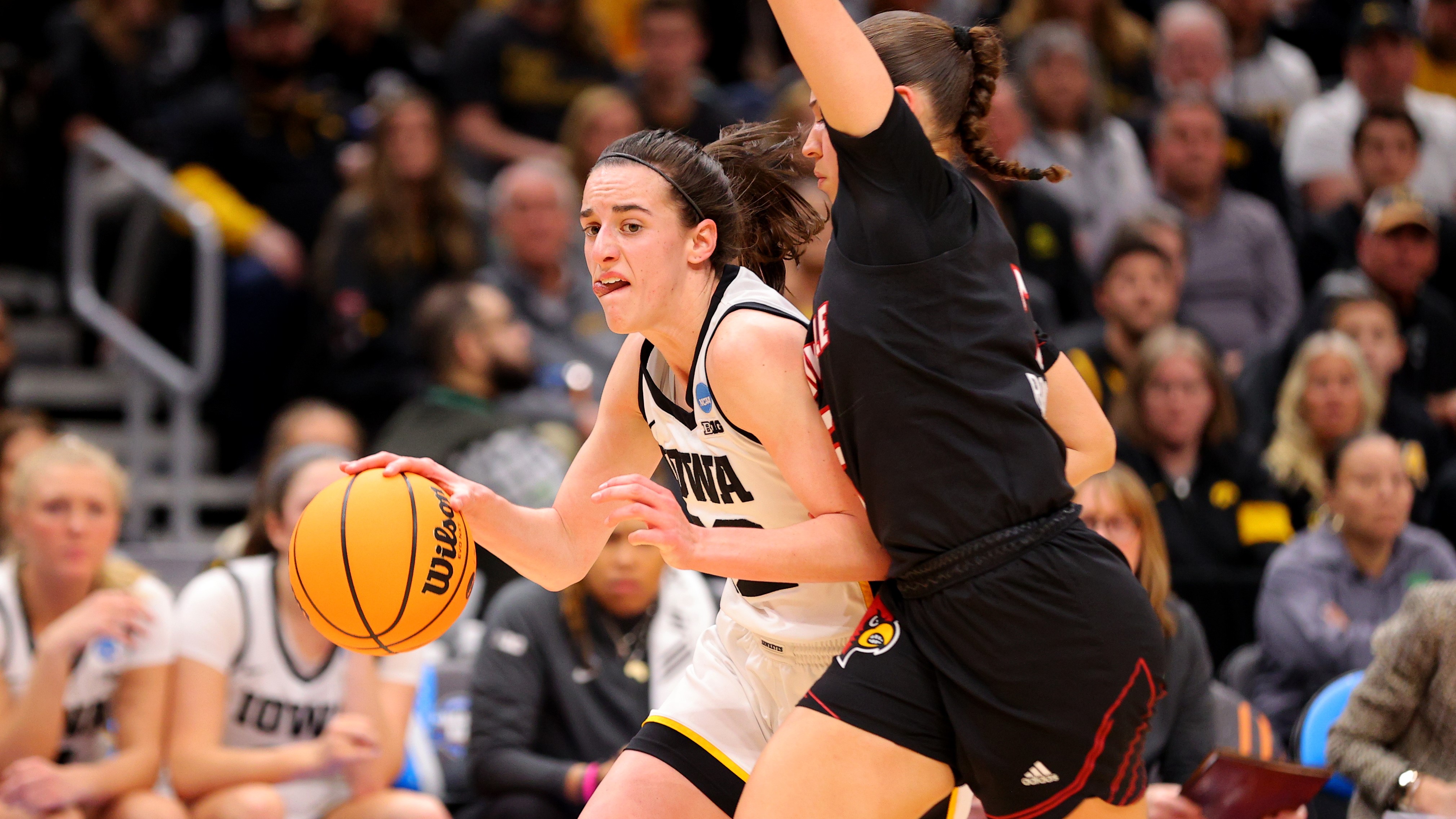
Horizon Sports & Experiences Signs Shark Beauty as Title Sponsor of College Basketball Women’s Champions Classic
By Jon Lafayette published
Caitlin Clark’s alma mater among top teams to compete in games to be broadcast by Fox in primetime

Inscape Working With Locality to Provide Viewing Data Across National and Local Linear TV and Streaming
By Jon Lafayette published
Brands will be able to determine incremental reach of campaigns

IAS Expands Media Quality Product to Identify Misinformation on YouTube
By Jon Lafayette published
Global advertisers can determine brand safety and suitability

Singing Janitor Wins ‘AGT’ Season 19
By Michael Malone published
Richard Goodall rode Golden Buzzer and some Journey classics to a million bucks

‘Agatha All Along’ Levitates to Top of TVision Power Score Rankings
By Jon Lafayette published
Emmys boost 'Shogun,' 'Hacks'
Anthem Sports & Entertainment To Buy Hollywood Suite, Canadian Channel Operator
By Jon Lafayette published
Acquisition would double Anthem's networks in Canada
The smarter way to stay on top of broadcasting and cable industry. Sign up below




